One of the most amazing things Dr. Jenn and I heard from our good clients in recent weeks and months as we’ve been catching them up with the status of our expansion project is, “When you’re all done, will we be allowed back inside?” The answer to this question is a resounding, “YES!!!” And as
read moreWe’re really excited to tell you construction has begun and our vision of an expanded building is unfolding before our very eyes! Our goal is to return to the in-person client experience of 2019. Of course all of this construction means further inconveniences for a time, and while we know a few of our clients
read moreIt is with great enthusiasm that we at Cuyahoga Falls Veterinary Clinic announce our expansion in 2022. 1999 brochure announcing CFVC’s pending move Cuyahoga Falls Veterinary Clinic began in 1981 with a small but loyal clientele. We have continued to grow since then in a multitude of ways, and because of wonderful clients like you,
read more[Veterinarian’s Note: In an effort to keep our clients informed while accounting for the tune-in factor, this reads much like some previous posts, with some updates sprinkled in.] There are hundreds of pet foods on the market, and everyone online and in the store aisles seems to have an opinion about what diet is best. We
read moreIt’s a poorly kept secret delivering veterinary medical services has become more of a challenge since the pandemic started. Nobody anticipated the world events would lead to a dramatic nationwide increase in animal adoption and pet ownership, but that’s exactly what we’ve observed and experienced. Surveys show this increase in pet ownership has been over
read moreWe’re sticking with our curbside model, and it’s not about Covid. For a variety of reasons, the demand for veterinary services has increased dramatically since March, 2020. This increased demand is a national as well as a local phenomena, and it has not slowed down. At times, the demand has outpaced our ability to adequately
read more
Covid and all of the changes to our personal lives and to our society that have come with it have been overwhelming. One cannot open a newspaper, turn on the radio or walk into a store without a blunt reminder that life in America has changed. Life at Cuyahoga Falls Veterinary Clinic has changed as
read moreWe’ve talked compliance before. It’s one of the greatest challenges in medicine, either on the human side or the veterinary side. We’ve also talked ProHeart6 before. This is the injectable heartworm preventive that also helps with intestinal parasite control, and it covers dogs for 6 months. ProHeart6 has been our go-to internal parasite control preventive
read more
You can’t turn on your television, visit your preferred social media platform, open a newspaper, or tune in your radio without hearing about COVID-19. We at the Cuyahoga Falls Veterinary Clinic want to address three facets of this topic. First, there is no evidence to suggest that our animals can acquire COVID-19 from us, nor
read more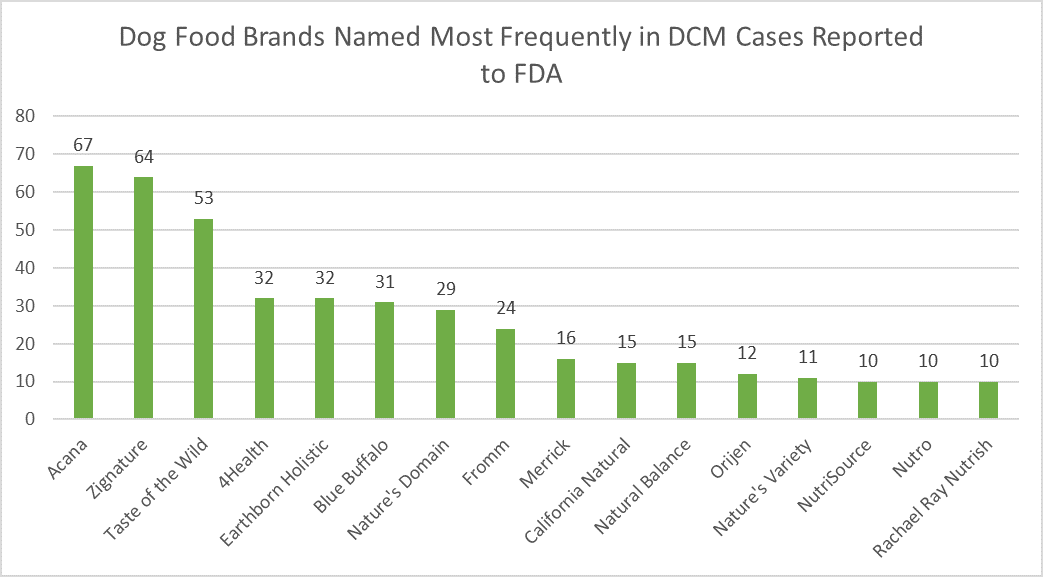
[Veterinarian’s Note: This reads much like our previous post, and includes updates from late June.] There are hundreds of pet foods on the market, and everyone online and in the store aisles seems to have an opinion about what diet is best. We at the Cuyahoga Falls Veterinary Clinic recommend providing diets manufactured by companies which
read more
